In earlier posts I have written about the performativity of manipulating materials. I have been ‘thinking’ with materials and recognising that they have their own inherent strategies. The performativity of my actions is coupled with the performativity of live/organic media.
For many years, I have been viewing science through the lens of ritualistic practice rooted in myth. Ritual is performative action with ordered ways of doing. Performance is an element of media that manifests itself in live-ness before an audience. Live-ness, for me, extends beyond the human body.
Sharing an interest in the ritual performativity of the live subject, I have been collaborating with performance artist and cultural engineer[M] Dudeck, whose work of inventing their own religion and mythology as art has merged with my interest in science as ritual rooted in Greek myth. Dialogue around shared themes led us to initiate Skin Bible, a mythological bio-art project that seeks to embed Dudeck’s queer mythos directly into polymer parchment.
With regard to my art practice, Skin Bible fits with over two decades of interest in the chimera; the fanciful artefact defined by contemporary biotechnology and ancient Greek culture. The term pertains to the hybrid monster, the transgenic organism and the illusion. Chimera is etymologically linked to khemia or alchemy – the magic art of transmutation or the fusion of disparate things to make new. Consider also new taxonomies for species, ecologies, gender politics, multiculturalism (socially engaged practice) and transdisciplinary studies. (Sperou, 2003, 2010)
Many artists working in the genre of bio-art, including myself, aim to decolonise culture away from Enlightenment perspectives, to be inclusive, demystified, feminist and subversive toward privilege of fully human over non-human. Consider also the politics of Terra Nullius (no body’s land), bio/techno hybridity, the hierarchies imposed on life and boundaries between disciplines. (Sperou, 2018)
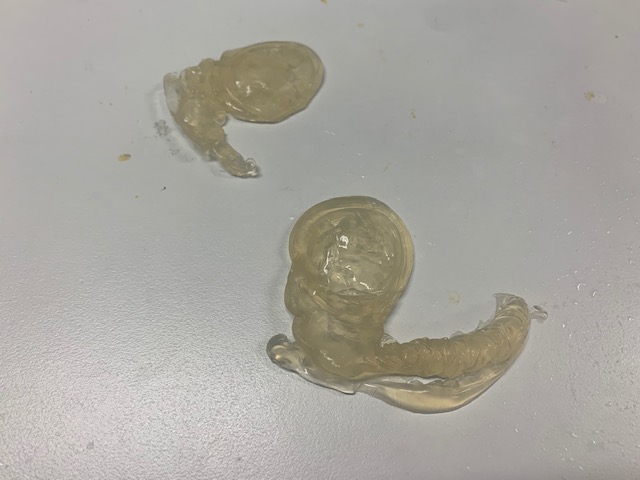
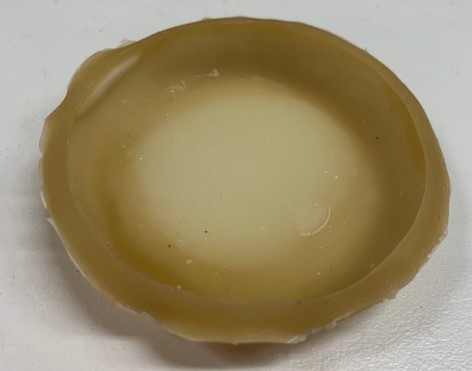
Marine algal polymers are promising biocompatible materials; a hybrid seaweed/skin polymer is envisioned for the Skin Bible. Inspired by anthropodermic bibliopegy (the tradition of binding books in human skin) and reliquaries (agalmata and anathēmata) a conventional alkaline alginate extraction method was adapted to create a leather.
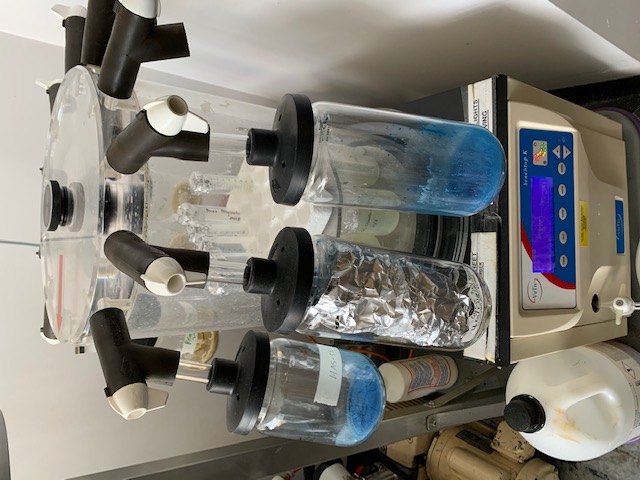
The complexity within the seaweed structure restricts cell wall degradation. Polysaccharides are natural polymers integrated within cell walls, which makes their extraction inherently difficult. We have under ecofriendly alkaline extraction achieved polysaccharides from species Durvillea potatorum (giant bull kelp) a brown marine macroalgae harvested from beach cast biomass from Beachport, South Australia (latitude: 37֯30’55”S; longitude 140֯4’17”E): a hotspot for marine algal diversity and endemic species. The seaweeds are rinsed and air-dried. The size of the kelp biomass is reduced by mechanical decortication to a fine powder. (Abraham et al, 2018)
Coupled with their low toxicity, biocompatibility, antimicrobial and antioxidant properties the polysaccharides are attractive for biomedical applications; wound dressings, tissue engineering as a copolymer for cell seeding & cell immobilisation scaffolds and 3D bioprinting.
Seaweed
Alkaline extraction (Stirring 2 h at 60֯c, with 28% NaCO₃)
Glycerol (Stirring 30 mins at 85֯c)
Pour and cast
Calcium chloride (spray)
(Conventional alkaline alginate extraction method)
References:
Sperou N, Chimera, the fanciful artefact defined by contemporary biotechnology and ancient Greek culture, Honours exegesis, 2003-2004
Sperou N, Verisimilitude: Biotechnology and Ancient Greek Narrative, Pandilovski M [Ed], Art in the Biotech Era, Experimental Art Foundation Inc, Australia, 2008
Sperou N, Chimera: material manipulations interfacing art, biotechnology and ancient Greek cultural paradigms, Khemia: Living Alchemy, exhibition catalogue essay, Royal Institution Australia, 2010
Abraham E et al, Optimisation of biorefinery production of alginate, fucoidan and laminarin from brown seaweed Durvillaea potatorum, www.elsevier.com/locate/algal, 2018
Sperou N. Uncertain creation; sympoietic system for art and science, Leonardo Journal, MIT Press, 2018, Leah Barclay et al, Abstracts from the Spectra 2018 Symposium: Environment and Bio-Science: (doi: accepted 10.1162/leon_a_1974)